From Workaround to Workflow
A complete redesign of a safety-critical clinical trial tool — rebuilt around workflows, not the technical constraints that created it.
Context
Rave Safety Gateway exists because of a technical gap. Medidata’s EDC platform — where clinical trial sites log all patient data, including adverse events — can’t natively transmit that data to external safety reporting systems.
RSG is the bridge. Study developers configure the field mappings so the right data flows to the right place in the right regulatory format (ICH E2B). Safety specialists then review and approve transmissions before they reach the safety system.
The product worked. But the experience had been built around the technical architecture — not around the people doing the work. That’s what we were there to change.
The Brief
The product team came to us with a request to modernise the interface. The framing was surface-level: update the visual design, clean things up, make it feel more current.
After a structured kickoff and six weeks of discovery — user interviews, research sessions, workflow analysis — it became clear that “modernise” was the wrong frame.
The problems weren’t cosmetic. They were navigational and cognitive. The tool exposed its internal architecture to users instead of abstracting it. The mental model it imposed didn’t match how study developers and safety specialists actually thought about their work.
We pushed back on redesigning things for the sake of redesigning them. We protected what users said was working, focused on what they said was hard, and defined scope around real pain — not product team assumptions.
“The product team wanted to redesign lots of small things for the sake of redesigning. They were somewhat neglecting what users actually wanted to change — and what didn’t need to be touched at all.”
The Key Insight
The most important thing we learned had nothing to do with UI patterns. It was about the rhythm of the work itself.
Study developers don’t complete a configuration mapping in one go. They work from ICH E2B specification documentation — a dense technical reference — that sits open alongside the tool the entire time. A full mapping session can take hours. Users routinely stop partway through and come back later, sometimes days later.
The old interface had no memory of this. There was no way to see at a glance what was done, what was left, or what state a section was in. Every return meant re-orienting from scratch, scanning the entire mapping to figure out where you’d left off.
The tool needed to hold the user’s place — not just technically, but visually. A persistent, sectioned navigation that doubled as a progress layer: not a percentage, but per-section completion status. So dropping back in after a break meant immediate orientation, not re-discovery.
Design Decisions
Three decisions, each tied directly to a research or structural finding.
01 — Navigation & IA
The old structure forced users into a flat “list of studies → drill in” pattern that gave no sense of position and no way to navigate across sections without losing context.
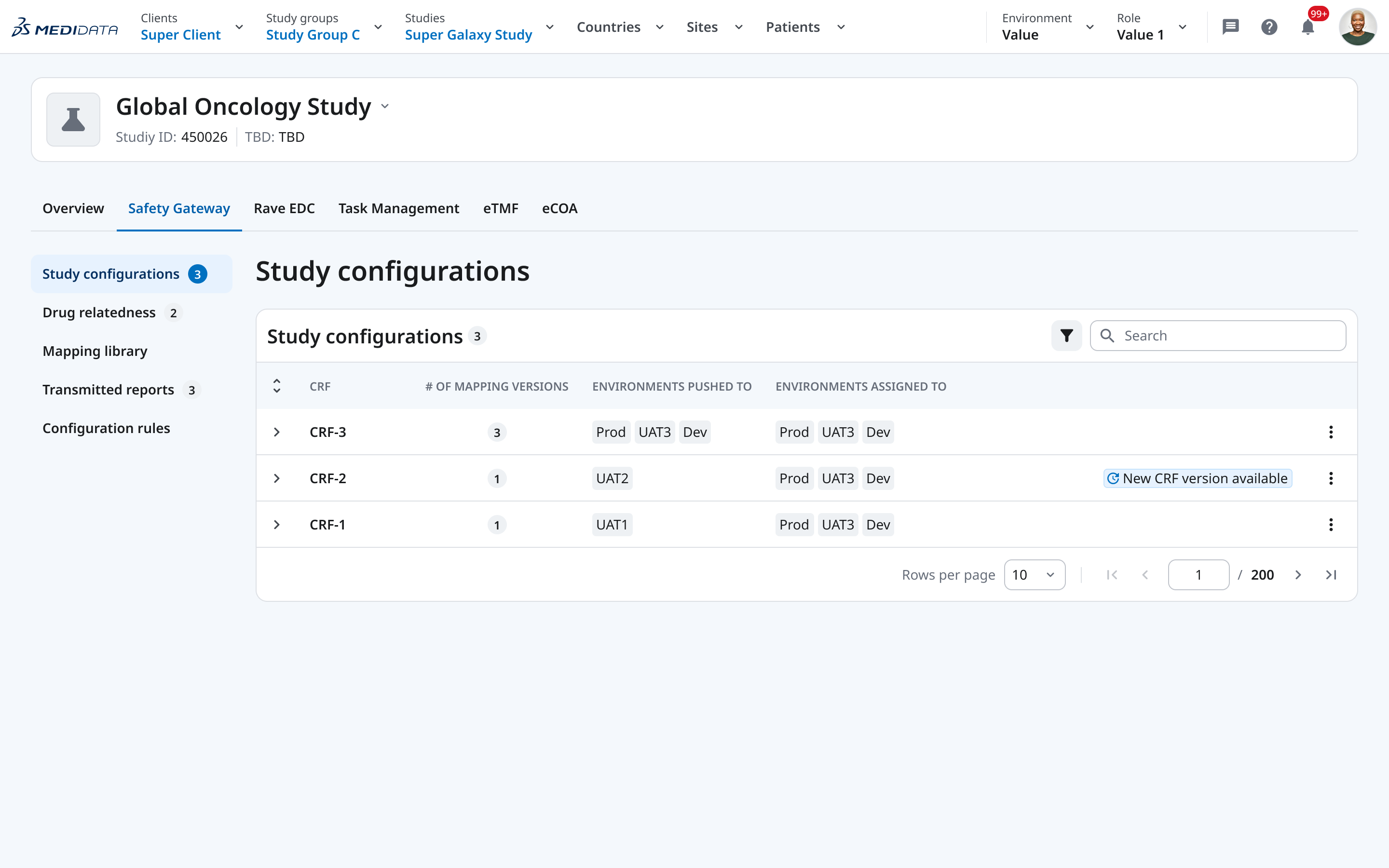
The redesign introduced a clear hierarchy: client → study group → study → Safety Gateway tab. Within a study, the left sidebar gives persistent access to all sections — Study configurations, Drug relatedness, Mapping library, Transmitted reports — so users always know where they are and can move without losing their place.
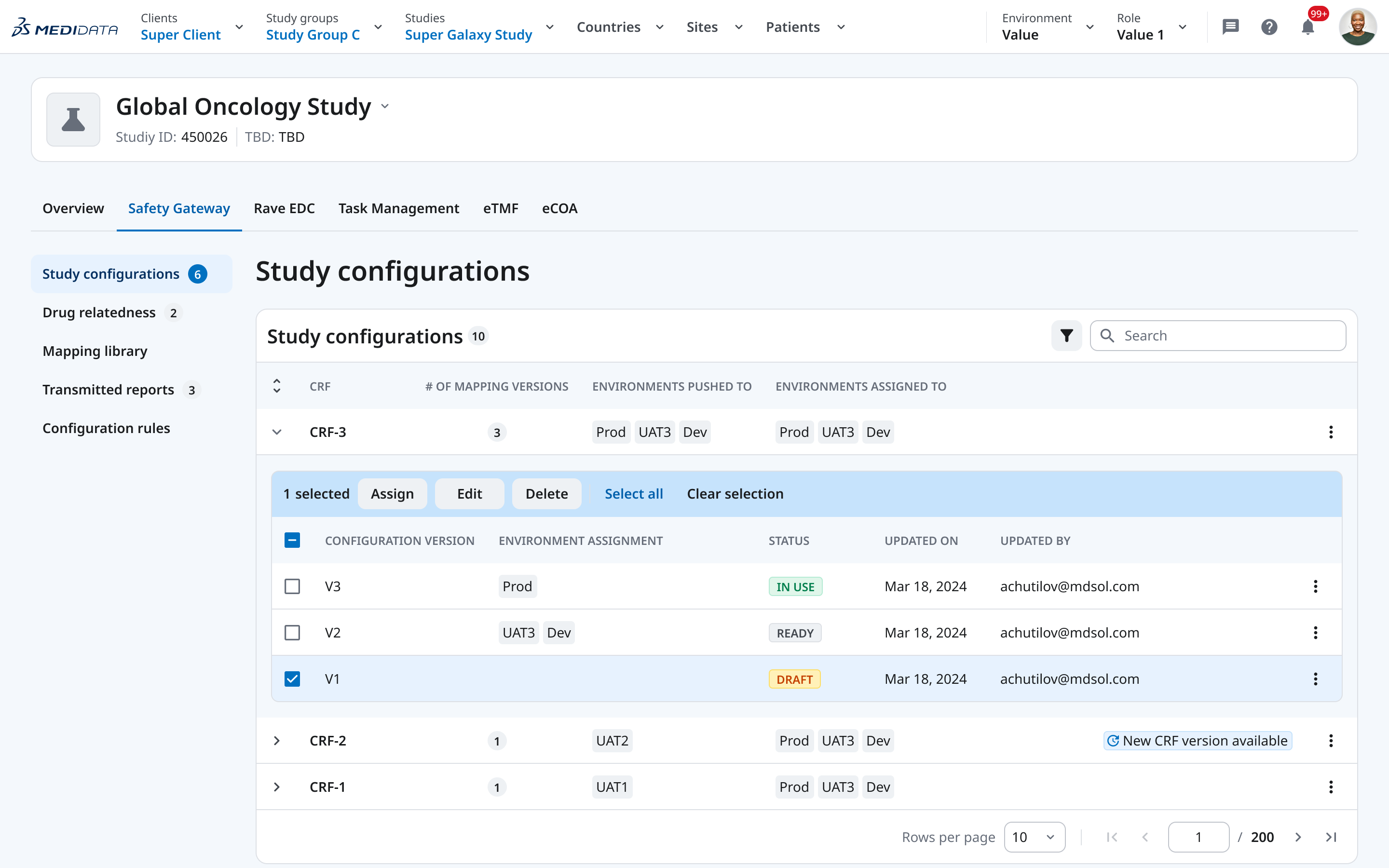
02 — Versioning Model
Before, the relationship between a configuration, its versions, and the environments it had been deployed to (Production, UAT, Dev) existed in the backend but was invisible in the UI. Users had to hold this model in their heads.
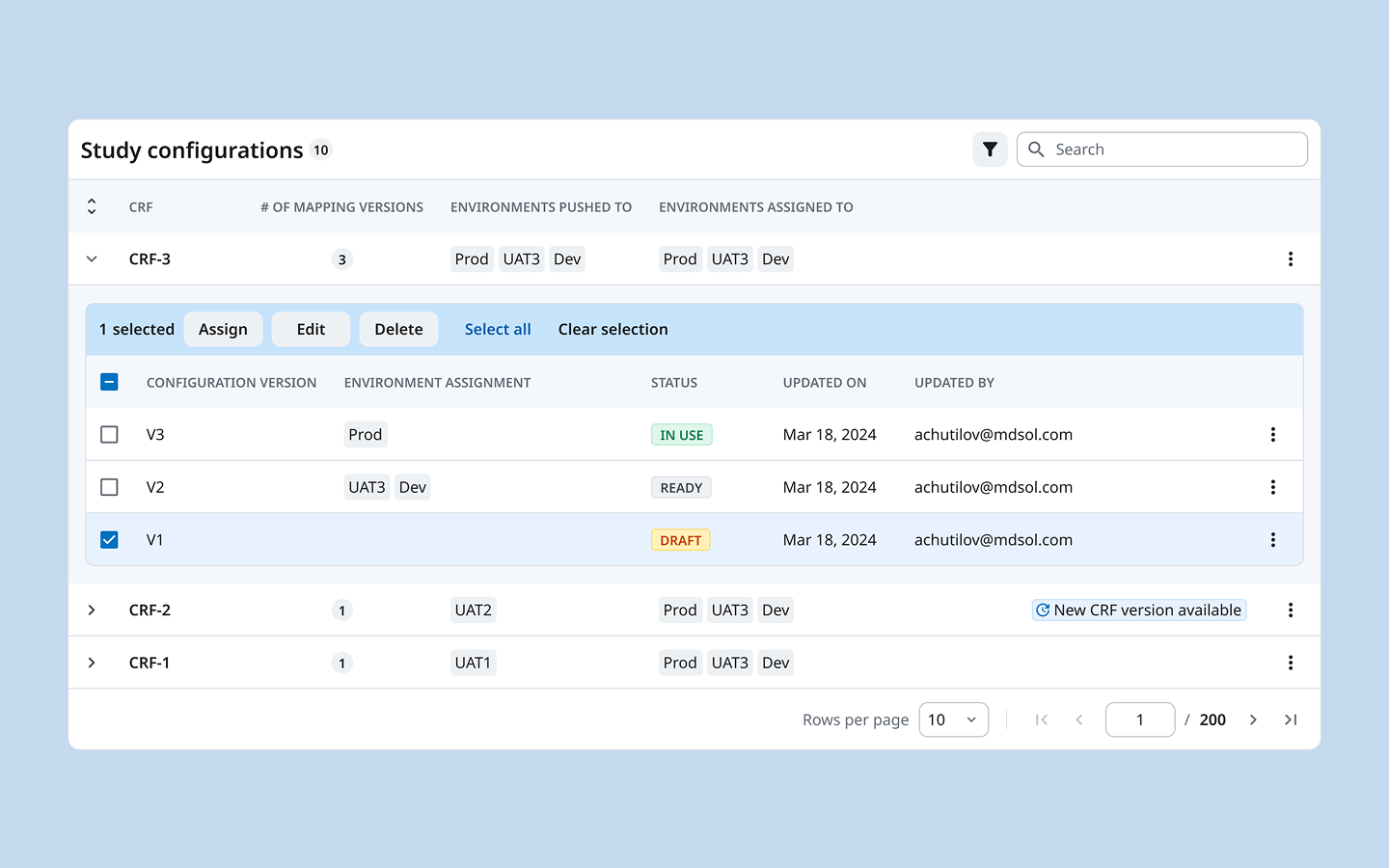
We introduced an explicit versioning structure: each CRF has named configuration versions (V1, V2, V3), each with a clear status (Draft / Ready / In Use), each explicitly assigned to one or more environments. The table makes the full picture readable in a single view — no clicking in to find out what’s live.
This replaced a cognitive load with a legible system. We designed this versioning model from scratch.
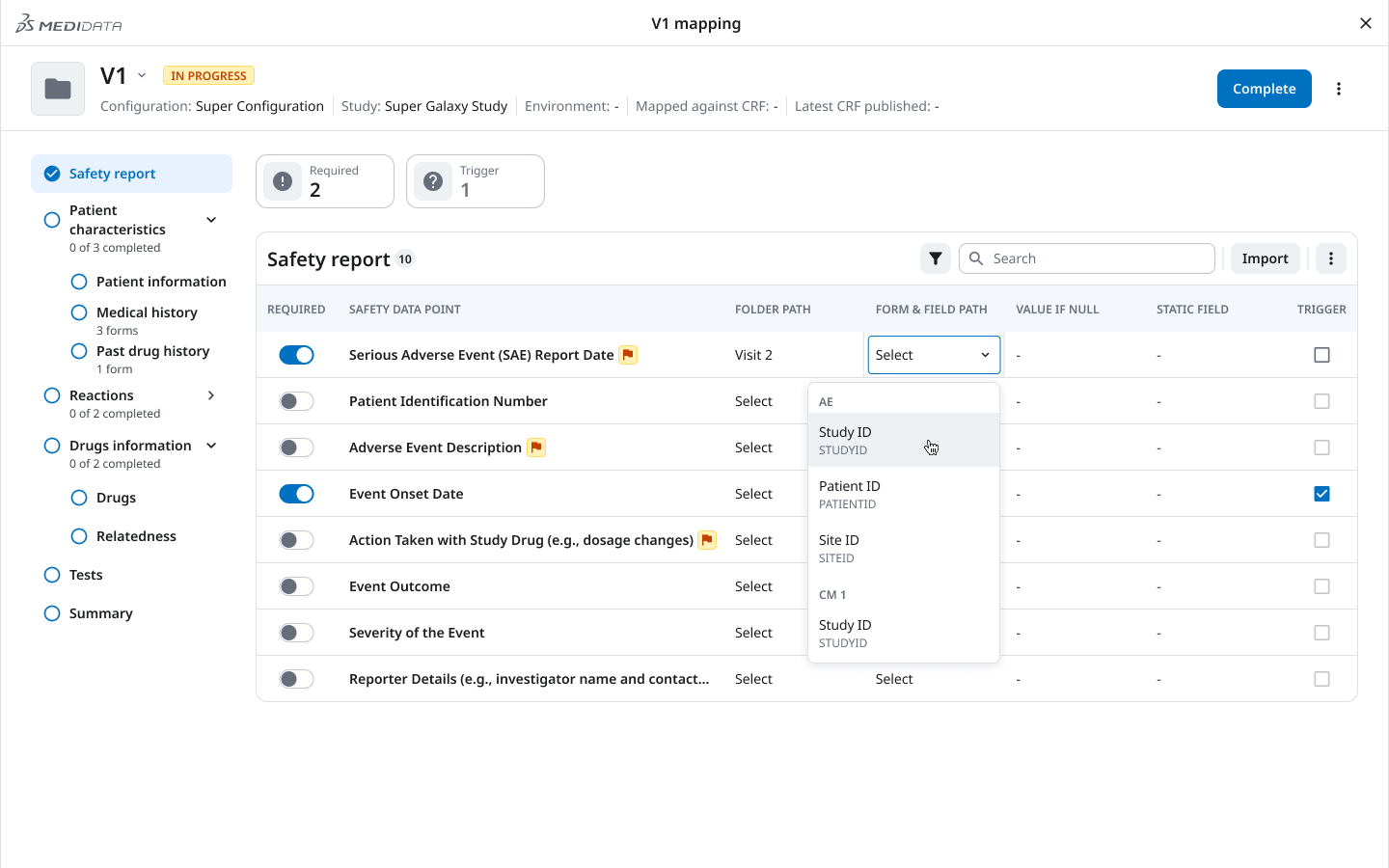
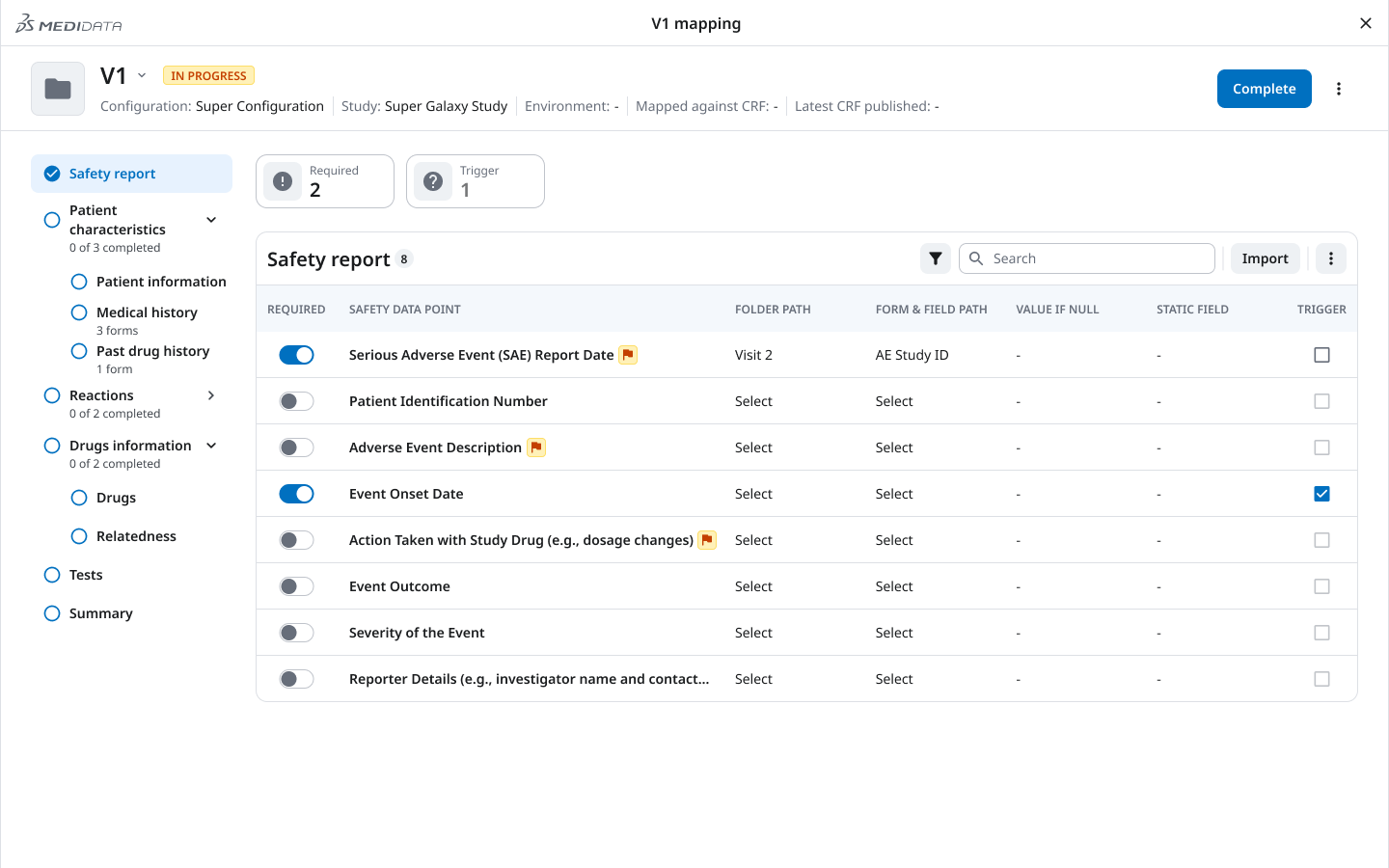
03 — Mapping Progress Sidebar
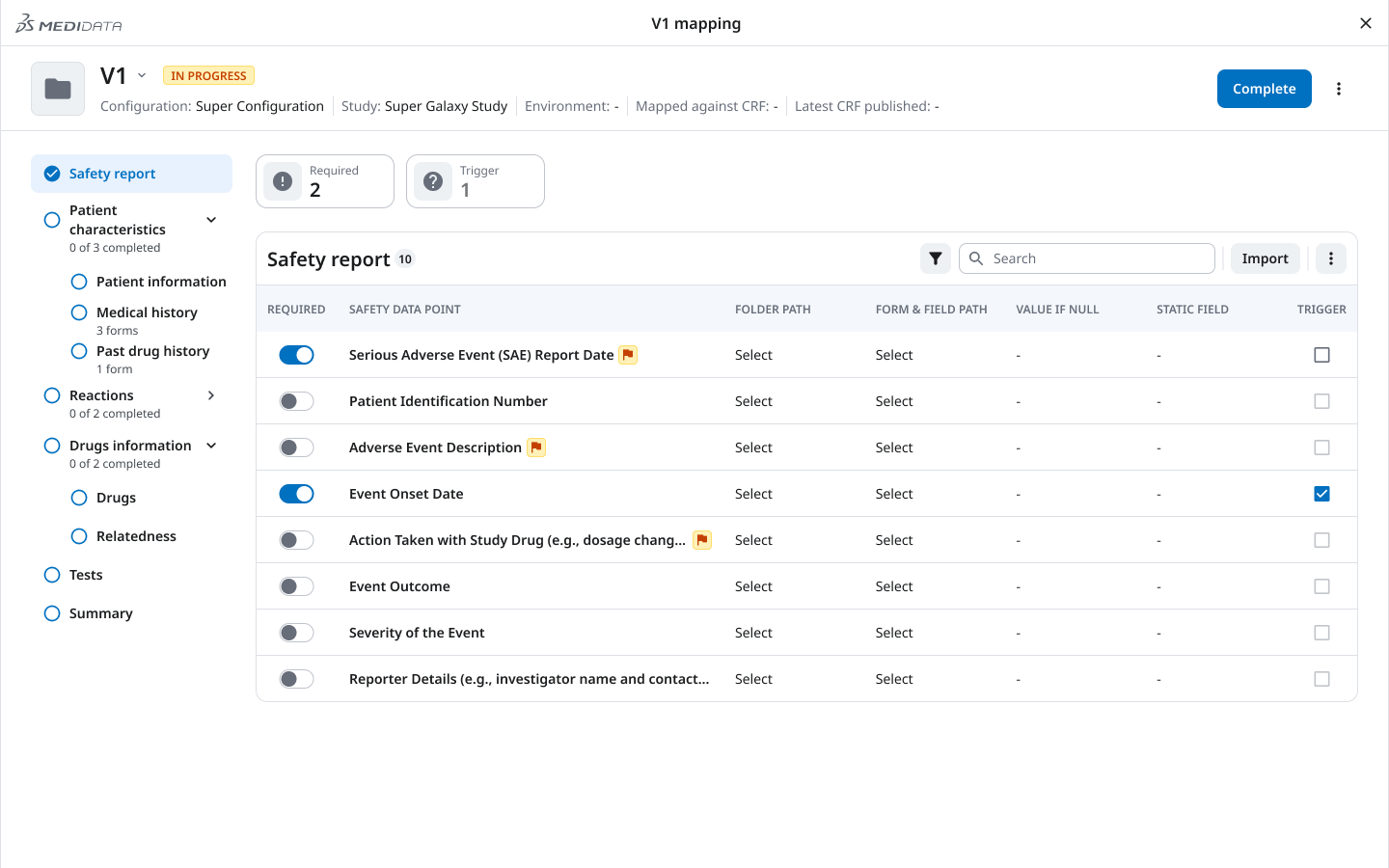
Directly shaped by the interrupted workflow insight: the mapping interface needed to tell users both where to go and what was done.
The persistent left sidebar shows each mapping section (Safety report, Patient characteristics, Medical history, Reactions, Drugs, Relatedness, Tests, Summary) alongside a completion indicator — “0 of 3 completed,” “2 of 2 completed.” Required fields are surfaced, not buried.
The result: drop back in after a break, open the sidebar, immediately know what’s done and what’s not. No scanning. No re-discovery.
More Screens
Outcome
“The whole process makes so much more sense now from a UX perspective.” — Usability test participant
The redesign was demoed at Medidata NEXT — the company’s flagship annual conference, attended by pharma, biotech, and CRO leaders from around the world. New client interest and bookings followed immediately after the product demo.